Denture Base Resin
- Bio Compatibility
- Skin Contact, Mucosal Membrane Contact
- Print Technology
- SLA
- Applications
- Full Dentures
- Compatible Printer
- Form 4B, Form 3B
- Colors
- Multiple Shades
- Flexural Strength
- 65.0 MPa
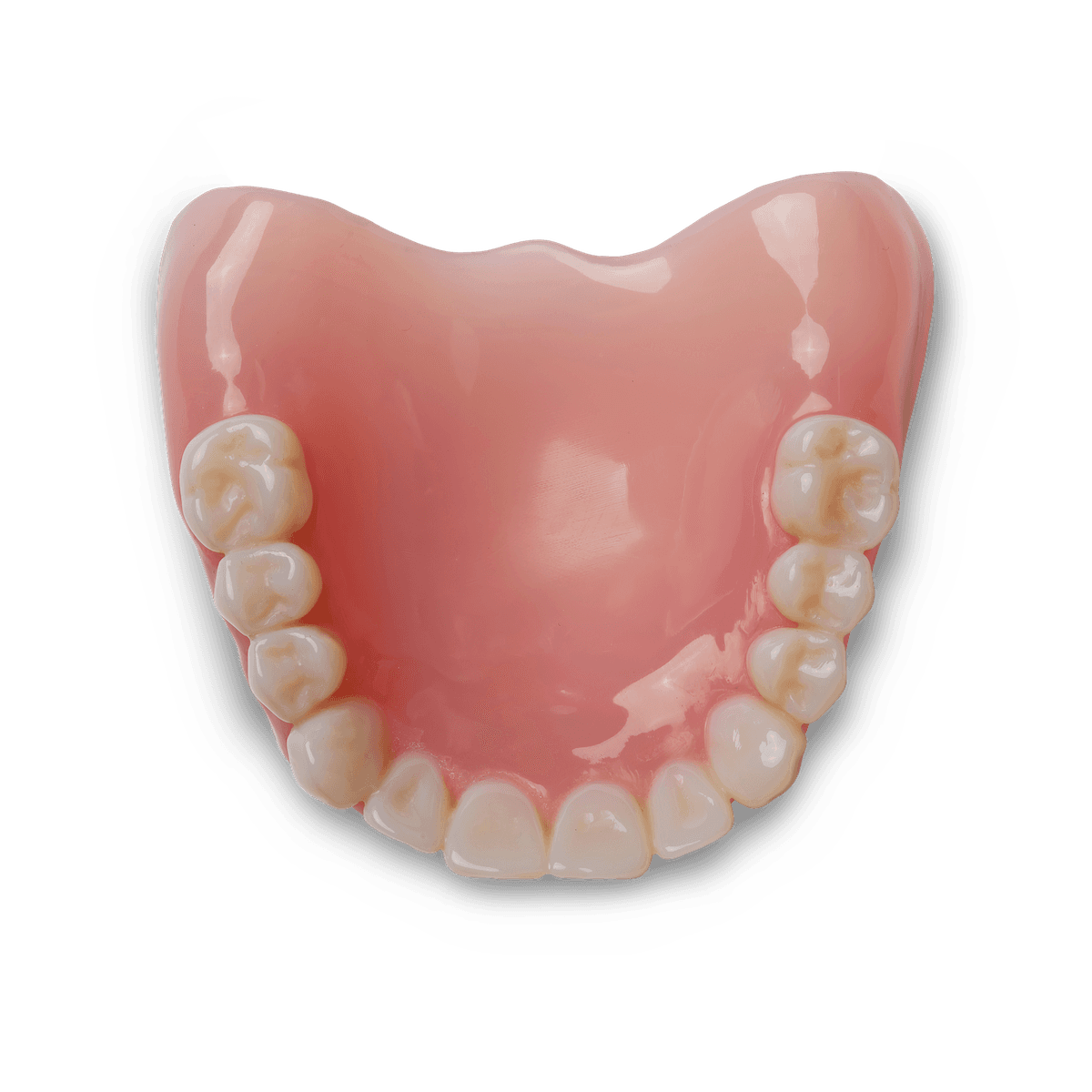
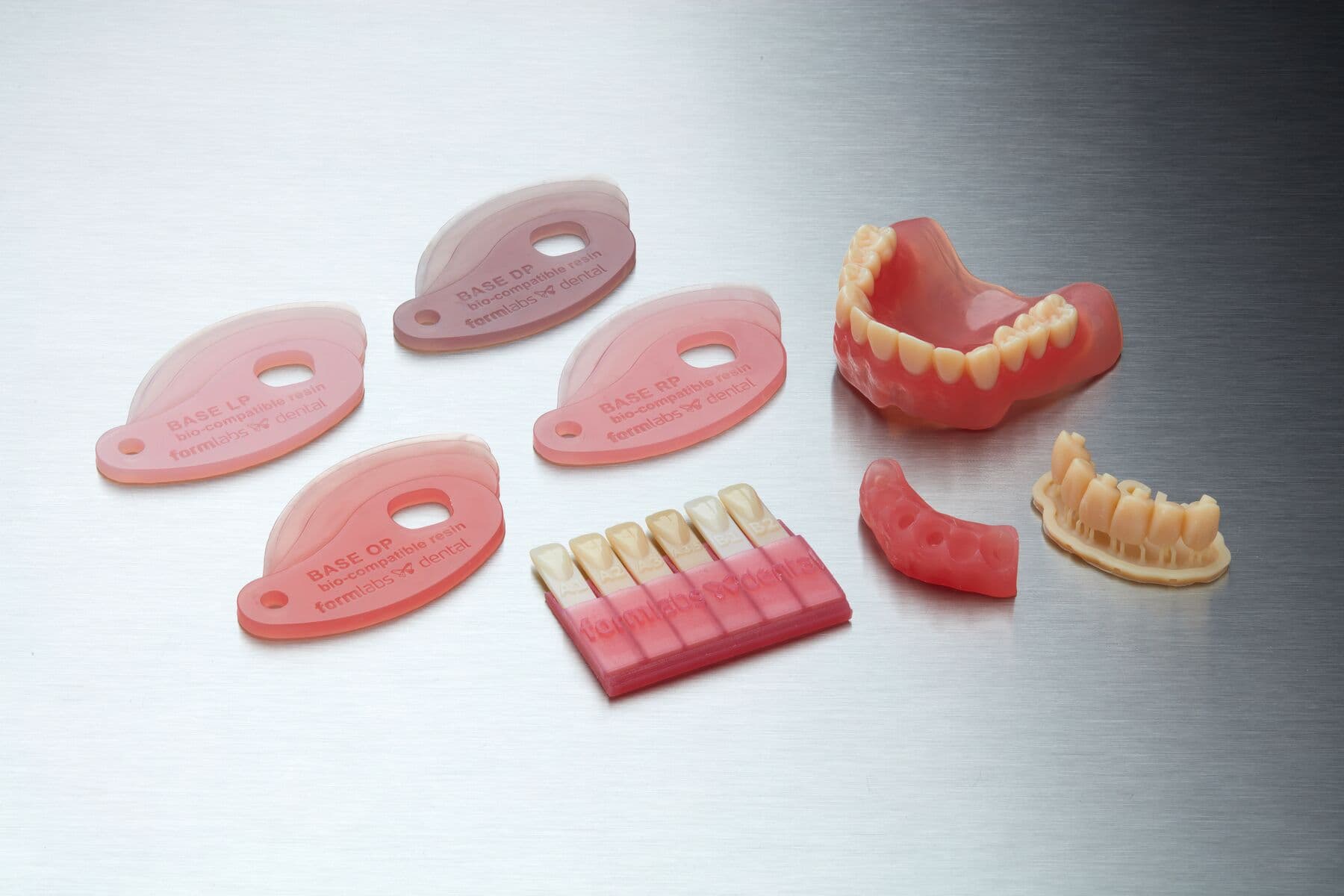
Denture Base Resin is a biocompatible material for producing long-lasting, wear-resistant denture bases and try-in dentures at a fraction of the cost of traditional methods. With four semi-translucent shades available, technicians can achieve lifelike gingiva with natural margins, whether with 3D printed denture teeth or acrylic cards.
Read our application guide to learn the complete digital workflow and best practices for making 3D printed dentures on your Formlabs 3D printer.
- Bio Compatibility
- Skin Contact, Mucosal Membrane Contact
- Print Technology
- SLA
- Applications
- Full Dentures
- Compatible Printer
- Form 4B, Form 3B
- Colors
- Multiple Shades
- Flexural Strength
- 65.0 MPa
Select Printer Series and Resin Volume
Resin Shade
Compatibility
- 3D Printer
- Form 3B, Form 2, Form 4B
- Build Platform
- Form 4 Build Platform, Form 4 Flex Build Platform, Form 3 Build Platform, Form 3 Build Platform 2, Form 3 Stainless Steel Build Platform, Form 3L Build Platform, Form 3L Build Platform 2L
- Tank
- Form 2 Resin Tank (PDMS), Form 3 Resin Tank V2.1, Form 4 Resin Tank
Denture Base Resin—
Why Choose Denture Base Resin?
Denture Base Resin enables dental professionals to produce 3D printed dentures accurately and reliably.
Semi-Translucent, Pink Base Shades
Provide lifelike gingiva with natural margins.
Strong and Wear-Resistant
Produce long-lasting dentures that won't break or wear down easily.
Repeatable and reliable
Reach an end result in fewer steps, start to finish, than traditional workflows.
Biocompatibility
Provide the patient and clinician the peace of mind they deserve.
Applications
Denture Base Resin is expanding access to digital dentures with an efficient, cost-effective manufacturing solution.
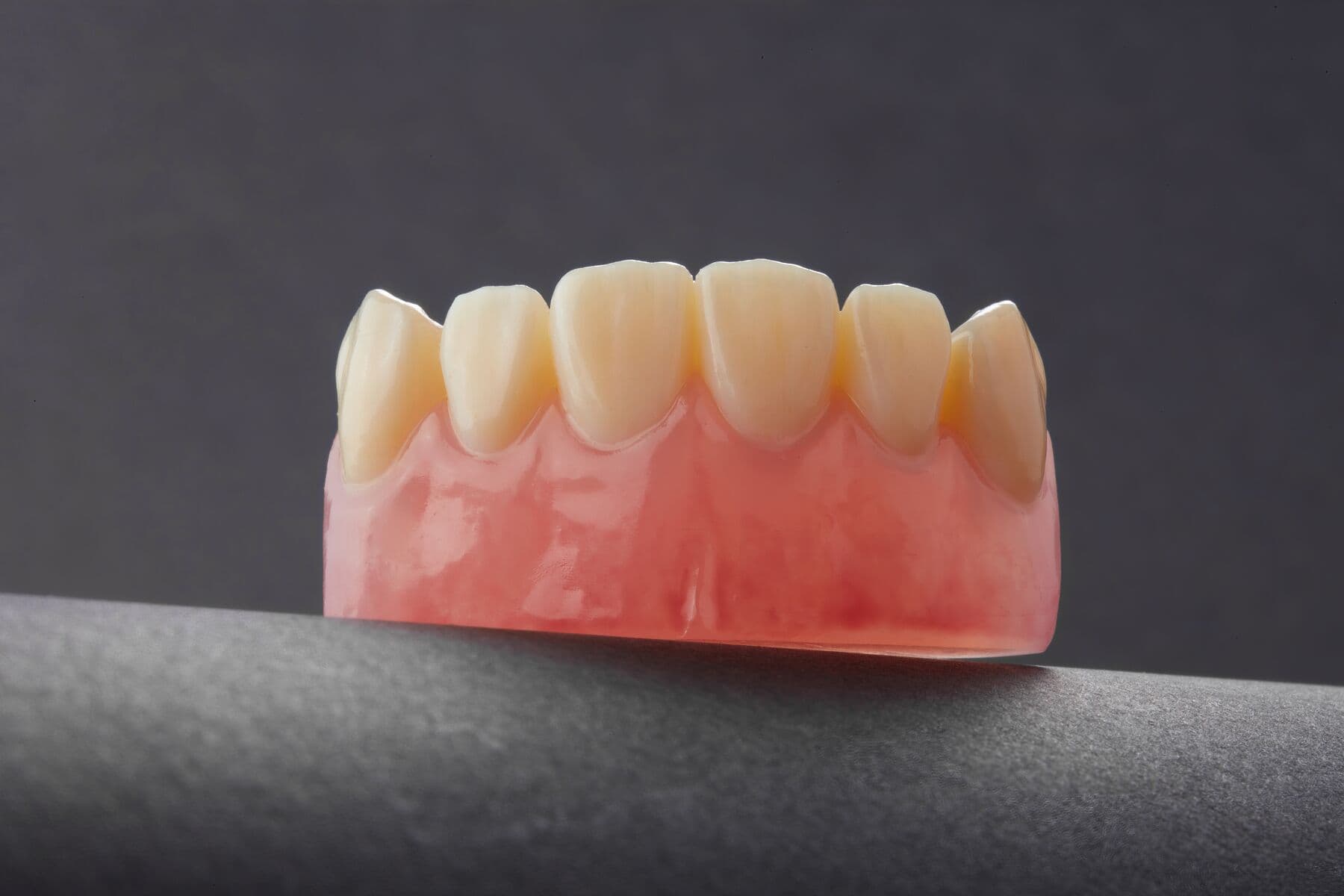
Final Dentures

Try-in Dentures

Manufactured in an ISO 13485-Certified Facility
Formlabs manufactures our Biomed and Medical Device Resins at our FDA-registered facility in Ohio. These materials are designed and manufactured within our robust Quality Management System that is ISO 13485 and EU MDR certified. A dedicated team of operators and quality assurance professionals makes the resins inside a certified ISO Class 8 clean room. All of our Medical Device Resins are appropriately registered with the FDA and CE marked according to the EU MDR.
Material Properties*
| Parameter |
Value |
|---|---|
|
Flexural Strength |
> 50 MPa |
|
Density |
1.15 g/cm 3 < X <1.25 g/cm3 |
* Material properties may vary based on part geometry, print orientation, print settings, and temperature. Data refers to post-cured properties obtained after exposing green parts to 108 watts each of Blue UV-A (315 – 400 nm), in a heated environment at 80 °C (140 °F) and 1hr, with six (6) 18W/78 lamps (Dulux blue UV-A).
Post-Processing
Washing
The foundational step in any SLA post-processing workflow is to remove excess resin on the surface of the parts through an alcohol wash. Please refer to the material's manufacturing guide/IFU for washing instructions.
Curing
For biocompatible materials, post-curing is necessary to achieve the safety standards determined by regulatory agencies. Please follow the material’s manufacturing guide/IFU for proper post-processing.